Инфанрикс Гекса особенно важна для младенцев и маленьких детей, у которых еще не сформирована
Принимая активные шаги для укрепления иммунной системы, вы можете значительно уменьшить риск заражения различными
Решение кроссвордов — это увлекательное занятие, которое может стать еще интереснее и проще, если
Толстой умер 20 ноября 1910 года на железнодорожной станции Астапово после бегства из дома
К люксовой и премиальной парфюмерии относятся широко известные дорогие ароматы, которые представлены в фирменных
В эпоху массового производства, персонализированные подарки и сувениры становятся все более ценными, и гравировка
Точность в медицинском переводе жизненно важна. Ошибка в переводе может привести к неправильной диагностике,
Выбор женского зонта не ограничивается только его внешним видом. Важно обращать внимание на качество
Вакцинация от бешенства (возбудитель — Rabies lyssavirus) является обязательной в виду особой опасности заболевания
Поход за украшениями в торгово-развлекательный центр (ТРЦ) может превратиться в настоящее приключение.
Весна 2024 года обещает быть яркой и новаторской в мире моды. Последние тенденции в
Военные тематические мероприятия представляют собой уникальный способ ознакомления с историей через погружение в атмосферу
Традиционная свадьба чаще всего включает в себя классические элементы: официальную церемонию бракосочетания, обмен клятвами,
Выбор гранитора зависит от нескольких факторов, включая объем напитков, который планируется продавать, и доступное
Пароконвектомат и духовой шкаф являются ключевым оборудованием в современной кулинарии, но многие задаются вопросом
При выборе аттракционов и реквизита для вашего праздника важно учитывать возраст участников, тематику мероприятия
Наркологическая клиника играет ключевую роль в процессе лечения и реабилитации людей, страдающих от наркотической
Онлайн-школы и курсы для будущих мам предлагают удобный и доступный способ получения важной и
Школьные экскурсии играют важную роль в образовательном процессе, предоставляя учащимся уникальные возможности для обучения
Это современный и эффективный метод удаления волос, обладающий высокой эффективностью, безопасностью и комфортом для
Сумки являются неотъемлемой частью повседневной жизни большинства людей. Они сочетают в себе практичность и
Ключевые инструменты в арсенале парикмахера включают в себя ножницы различных типов, расчески, щетки, машинки
Когда в доме есть дети, бардак становится частью повседневной рутины. Разбросанные игрушки, разорванные книги,
Новый год – время волшебства и подарков, особенно для детей. Детские новогодние подарки сладкие
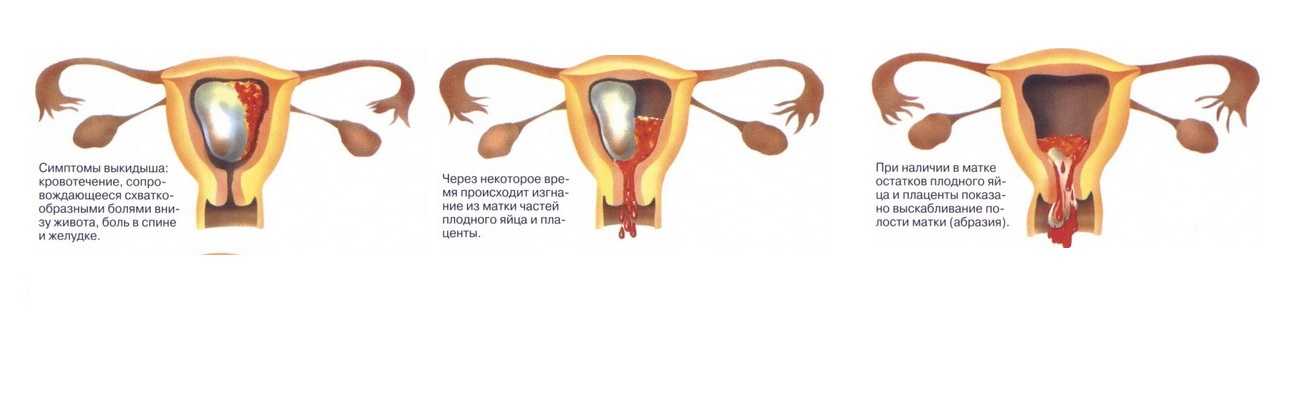
Важным аспектом ухода за своим здоровьем является регулярное посещение врачей-специалистов, включая гинеколога. В данной
Раньше об этом старались не говорить, но сейчас знаменитости без стеснения откровенничают на тему собственной
Сегодня расскажем о девочках, которые невероятно похожи на своих бабушек – знаменитых и не